About Us
Located in London, Pharma Design Limited is a service provider supporting clinical development aimed at innovative therapies. From CTA applications to MAA and HTA dossier preparation, Pharma Design is here to support sponsors of medicinal products intended for human use. Our customers achieve early access to European Markets by implementing a good strategy from the early stages of clinical development. Our pragmatic and flexible model have made Pharma Design Limited a trusted partner for many pharmaceutical organisations and biotechs seeking entry into European markets.
PhD Limited was founded in 2009 by Simon Ruini and a group of clinical, regulatory, market access and safety professionals willing to combine experiences and knowledge from different backgrounds to create an integrated service provider. Today, Pharma Design Limited is an efficient and reliable partner to Sponsors of pharmaceutical product development Worldwide.
From CTA applications to MAA and HTA dossier submission, Pharma Design supports the pharmaceutical development of medicinal products intended for human use. We help our customers achieve full access to European Markets by preparing a good strategy from the early stages of clinical development.
We have pioneered ATMP access within early stage pilot applications offered by EMA as of late 2025. We have supported new drug developers obtaining CTA approvals in Europe. We have helped sponsors obtain Marketing Authorisations in Europe in several different therapeutic areas.

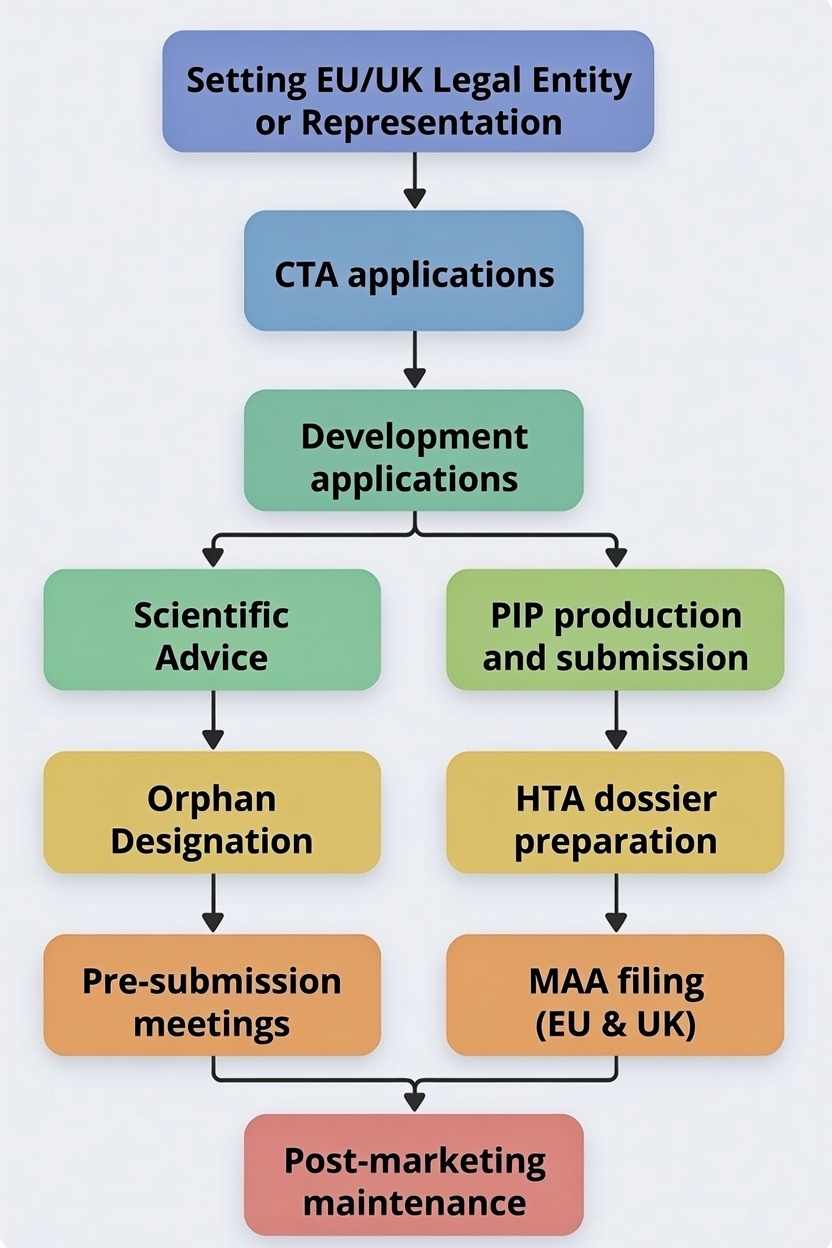
We are a single-shop solutions provider who is capable of integrating Regulatory, Clinical, HTA information along the development process into a successful submission.
Our technical consultants can work with you, individually or in small teams, delivering a first-class professional service as well as offering expert training. This is achieved through knowing and working with our pharmaceutical consultants and choosing the optimum skill set and experience for each individual project.
We can support your development form the earliest stages of research. Interacting early with Regulatory Agencies and Payers in Europe is not a waste of efforts. The need for evidence generation ahead of phase 3 studies together with long-term resource planning supports earlier interaction. It’s pure logic. This change is being translated into legislative changes that mandate Regulatory planning earlier in the game. You will not be granted PRIME in EU or ILAP in the UK if you do not apply before the start of phase 3 pivotal studies. It’s that simple!
We specialise in:
Market Research, Meta-analysis, Pricing Dossier, HTA
CTA Submission, Centralised MAA, Scientific Advice, License Maintenance
Benefit/Risk Assessment, PBRER, DSUR, RMP
PV Audit, DDPS, PSMF, PV Inspection preparation
Simply call us to discuss your project needs or send an e-mail, and within 48 hours, we can provide you with advice on the feasibility of your request and our ability to help. We aim to provide an estimate of project costs and a proposal within six working days.
Phone +44(0)755 798 3864 today to speak to our team.
Privacy Policy
Pharma Design Ltd (“we”, “us” or “our”) is committed to protecting your privacy and handling your personal data in accordance with the UK General Data Protection Regulation (UK GDPR) and the Data Protection Act 2018.
Information We Collect
When you visit our website, contact us via the enquiry form, email, or telephone, or engage with our regulatory, market access, or consulting services, we may collect the following personal data:
- Contact details (name, email address, telephone number, company name)
- Professional information (job title, company affiliation)
- Any information you voluntarily provide in messages, enquiry forms, or during consultations
- Technical data (IP address, browser type, pages visited) collected via cookies for website analytics and performance
How We Use Your Personal Data
We process your personal data for the following purposes:
- To respond to your enquiries and provide regulatory or market access consulting services
- To manage our client and business relationships
- To improve our website and services (using anonymised analytics)
- To comply with legal and regulatory obligations
We rely on the following lawful bases for processing:
- Legitimate interests (e.g., responding to enquiries and providing services)
- Contract (where we are providing services to you or your organisation)
- Legal obligation (where required by law)
Sharing Your Data
We may share your personal data with trusted third-party service providers (such as IT hosts, email service providers, or cloud storage providers) who assist us in operating our business. All such providers are bound by appropriate data processing agreements.We do not sell your personal data to third parties.International TransfersIf any data is transferred outside the UK, we ensure appropriate safeguards are in place (e.g., UK-approved transfer mechanisms).Your RightsUnder UK GDPR, you have the right to:
- Access the personal data we hold about you
- Request correction or deletion of your data
- Object to or restrict certain processing
- Request data portability (where applicable)
To exercise any of these rights, please contact us at [email protected] and Website AnalyticsOur website uses essential cookies and may use analytics cookies to understand how visitors use the site. You can manage your cookie preferences through your browser settings.Data Retention
We retain your personal data only for as long as necessary to fulfill the purposes for which it was collected, or as required by law.
Contact Us
If you have any questions about this Privacy Policy or how we handle your personal data, please contact:Pharma Design Ltd
Email: [email protected] Last updated: March 2026
