The EU Pharmaceutical Legislation reform (commonly called the Pharma Package) represents the most significant overhaul of EU medicines rules in over 20 years.
It consists of a new Directive (replacing Directive 2001/83/EC) and a new Regulation (replacing Regulation (EC) No 726/2004), along with amendments to related laws on paediatric medicines, orphan drugs, and advanced therapies.
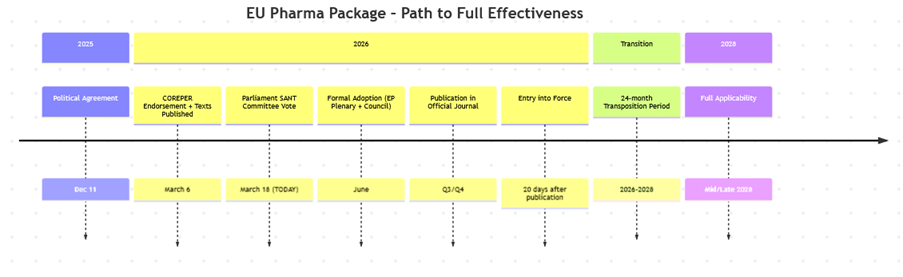
Current Status (as of March 18, 2026)
- Political agreement was reached between the European Commission, Parliament, and Council on December 11, 2025.
- Compromise texts were endorsed by COREPER I (Council) on March 6, 2026, and published.
- The Parliament’s ENVI/SANT Committee is voting on March 18, 2026 (today).
- Formal adoption by Parliament and Council is expected in summer 2026 (potentially by June), followed by translation into all EU languages and publication in the Official Journal.
- Entry into force is anticipated in 2026, with a 2-year transition period leading to full applicability around 2028 (e.g., 24-month transposition for the Directive).
The final texts closely reflect the December 2025 compromise, focusing on faster access, innovation incentives, supply security, and competitiveness while maintaining high safety standards.
Key Proposed Changes (Main Reforms from the Compromise Texts)
- Accelerated Regulatory Processes:
- Centralized marketing authorisation assessment timelines reduced from 210 days to 180 days.
- Simplified EMA scientific committee structure (from five to two main committees: CHMP and PRAC).
- Unlimited validity for marketing authorisations (no renewal needed).
- Promotion of electronic submissions and structured data formats.
- Regulatory Data Protection (RDP) and Market Protection (MP):
- Baseline: 8 years of data protection (no change) + 1 year of market protection (reduced from 2 years) → total 8+1 years.
- Modular extensions (up to a cap of 11 years total protection):
- +1 year for products addressing unmet medical needs (UMN).
- +1 year for products with comparative clinical trials, trials in multiple Member States, and EU filing within 90 days of first non-EU submission.
- Additional incentives for novel antimicrobials, breakthrough therapies, or specific conditions (e.g., orphan drugs).
- Orphan Medicines and Paediatric Incentives:
- Orphan market exclusivity reduced from 10 years to 9 years baseline.
- Possible +2-year extension for breakthrough orphan products.
- More flexible, iterative approach to Paediatric Investigation Plans (PIPs), with updates during development.
- Medicine Shortages and Supply Chain Resilience:
- Stronger obligations for MAHs to prevent and mitigate shortages (e.g., better monitoring, notification, and coordination).
- Launch obligations in all Member States (or justification for delays).
- Enhanced environmental risk assessments (e.g., for antimicrobials and AMR selection).
- Innovation and Access Boosts:
- Expanded Bolar exemption for generics/biosimilars (earlier development/testing).
- Stronger incentives for novel products, breakthrough therapies, and secure testing environments.
- Faster access to medicines across the EU, with emphasis on equitable availability regardless of country.
- Other Notable Elements:
- Streamlined procedures and reduced red tape for lifecycle management.
- Clearer rules on advertising, fiscal imports, and compounding.
- Sustainability integration (e.g., environmental considerations in approvals).
Overall Objectives
The reform aims to:
- Deliver timely and equitable access to safe, effective, affordable medicines.
- Boost EU pharmaceutical competitiveness and innovation.
- Strengthen supply security and reduce shortages.
- Support strategic autonomy in healthcare.
