Strategic Synergy: Integrating Regulatory Affairs & HTA
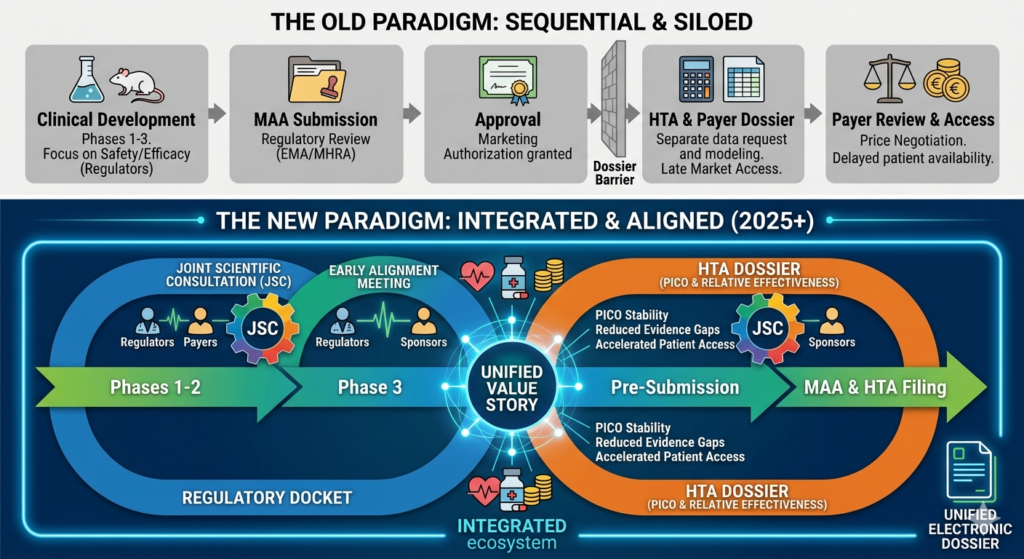
In the evolving landscapes of the EU and UK, the traditional “siloed” approach to drug development is no longer viable. With the implementation of the EU HTA Regulation (HTAR) and the UK’s increasingly collaborative MHRA/NICE Aligned Pathway, the bridge between Marketing Authorization (MA) and Market Access (HTA) has narrowed.
At Pharma Design, we specialize in the strategic integration of Regulatory and Health Technology Assessment (HTA) workstreams. We don’t just help you get a label; we help you get a price and a place in the market.
Why is Integration the New Standard?
New legislative frameworks in EU and UK now demand that clinical evidence satisfies two different masters simultaneously:
- Regulators (EMA/MHRA): Focus on Benefit-Risk, Safety, and Quality.
- HTA Bodies (NCPE, NICE, HAS, etc.): Focus on Relative Effectiveness and Cost-Utility.
By aligning these requirements during the development phase, we eliminate the “evidence gap” that often leads to delays in reimbursement or restrictive market access.
Our Integrated Service Pillars
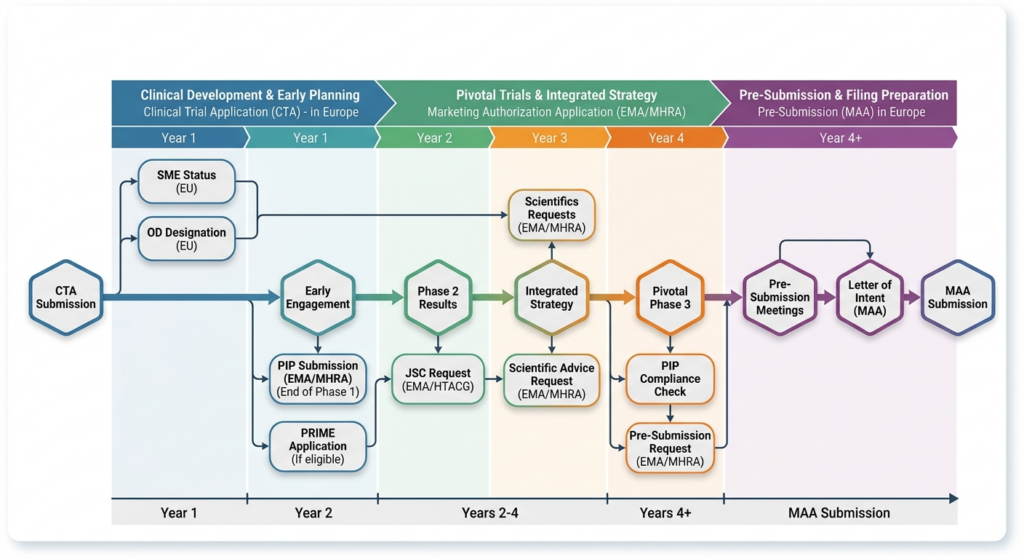
1. Strategic Alignment of Early Milestones
We synchronize your regulatory milestones with HTA requirements to ensure every data point serves a dual purpose.
- Scientific Advice: Coordinating joint clinical advice to ensure endpoints satisfy both regulatory scrutiny and payer value assessment.
- Pediatric Investigation Plans (PIP): Ensuring pediatric data is robust enough for HTA evaluation.
- Expedited Pathways: Navigating PRIME (EU) and PIM/ILAP (UK) designations while front-loading HTA dossier preparation.
2. Joint Scientific Consultations (JSC)
Pharma Design has experience facilitating JSC meetings with the EMA and HTA bodies. We manage the complex preparation required for these meetings, helping you secure consensus on:
- Choice of comparator.
- Relevant patient-reported outcomes (PROs).
- Sub-group analysis requirements.
3. The Aligned Pathway (UK Focus)
Leveraging our deep expertise in the UK market, we assist clients in navigating the Innovative Licensing and Access Pathway (ILAP). We help bridge the gap between the MHRA’s safety review and NICE/SMC’s value assessment, ensuring a seamless transition from “Authorized” to “Available.”
4. Evidence Generation & Gap Analysis
We perform rigorous “stress tests” on your Phase II/III protocols to identify areas where HTA bodies might find the evidence insufficient, allowing for course correction long before the MAA filing.
The Pharma Design Advantage
- Legislative Fluency: We stay ahead of the latest EU HTAR requirements, ensuring your dossiers are compliant with the new mandatory joint clinical assessments.
- Reduced Time-to-Patient: By aligning HTA preparation with the MAA filing, we significantly reduce the “dead time” between regulatory approval and national reimbursement.
- Risk Mitigation: We identify conflicting requirements early, preventing the need for costly post-marketing trials.
“Regulatory approval is a milestone, but patient access is the goal. We design the path to achieve both.”
Ready to Align Your Strategy?
The complexity of the EU and UK markets requires a partner who understands the interplay between policy and science.

Establish a European Legal Entity and Register with Agency Portals.
If you are preparing for a Marketing Authorisation in Europe you will need to establish a legal presence in Europe (EU, UK, Switzerland). You will then need to register your local representatives (we can represent you if you wish) and your Product with Agency portals like EMA OMS, XEVMPD, MHRA Gateway and SwissMedic. Pharma Design Limited can assist you in setting up your European legal representation and to register your Products correctly in relevant portals, so that you will be officially recognised as a developer of new medicines and be contactable by Regulatory Authorities for any communication.
Our Values
The value we bring to pharmaceutical companies falls into four main areas:
- Objective, impartial advice tailored to your needs
- Help cut your development costs
- Help reduce your time to market
- Keep your drug on the market

